Two years ago, immunologist and medical-publishing entrepreneur Leslie Norins offered to award US$1 million of his own money to any scientist who could prove that Alzheimer’s disease was caused by a germ.
The theory that an infection might cause this form of dementia has been rumbling for decades on the fringes of neuroscience research. The majority of Alzheimer’s researchers, backed by a huge volume of evidence, think instead that the key culprits are sticky molecules in the brain called amyloids, which clump into plaques and cause inflammation, killing neurons.
Norins wanted to reward work that would make the infection idea more persuasive. The amyloid hypothesis has become “the one acceptable and supportable belief of the Established Church of Conventional Wisdom”, says Norins. “The few pioneers who did look at microbes and published papers were ridiculed or ignored.”
In large part, this was because some early proponents of the infection theory saw it as a replacement for the amyloid hypothesis. But some recent research has provided intriguing hints that the two ideas could fit together — that infection could seed some cases of Alzheimer’s disease by triggering the production of amyloid clumps.
The data hint at a radical role for amyloid in neurons. Instead of just being a toxic waste product, amyloid might have an important job of its own: helping to protect the brain from infection. But age or genetics can interrupt the checks and balances in the system, turning amyloid from defender into villain.
And that idea suggests new avenues to explore for potential therapies. To test the theory further, scientists are now developing animal models that mimic Alzheimer’s disease more closely. “We are taking the ideas seriously,” says neuroscientist Bart de Strooper, director of the UK Dementia Research Institute at University College London.
Choked by clumps
The amyloid hypothesis holds that Alzheimer’s results from a build-up of sticky, soluble proteins — amyloid-β peptides — in the spaces between brain cells. These peptides are cleaved from another protein embedded in the membranes of neurons. Once floating free, they clump together into larger structures which, if not cleared efficiently enough by special enzymes, aggregate into plaques. The plaques then trigger a deadly cascade: they provoke neuroinflammation and spawn bundles of stringy proteins called tau tangles. Faced with this litany of insults, neurons die.
Critics of the hypothesis note that the brains of many people who did not have Alzheimer’s disease have been shown to contain plaques on post-mortem. And they point to the failure of many clinical trials of treatments designed to dissolve amyloid plaques, none of which has slowed the disease. Researchers who support the amyloid theory counter that although the density of the plaques varies a lot between individuals, the density of tau tangles that they trigger correlates tightly with the severity of disease. And clinical trials probably failed, they say, because the treatments were given too late in the course of disease.
They also have strong evidence on their side. There are certain rare and aggressive forms of Alzheimer’s disease that emerge early — between the ages of 30 and 60 — and run in families; these conditions are caused by mutations in genes that govern the amyloid-making process and inflammation in the brain. Scores of other genes have been associated with the risk of the more common late-onset form of the disease. Several code for proteins that comprise elements of the amyloid cascade, and some are involved in the innate immune system — a group of mechanisms that activate quickly to prevent the spread of pathogens in the body, and which drive inflammation.
Agents of infection
Researchers hoping to test the infection hypothesis have gone hunting for microbes in thousands of post-mortem brains from people with Alzheimer’s. In many, they have found them. “But these studies only show correlations which may have explanations that have nothing to do with mechanisms,” says de Strooper.
Ruth Itzhaki, a biophysicist at the University of Manchester, UK, who reported observations of herpes simplex virus 1 (HSV1) in post-mortem Alzheimer’s brains in the 1990s1, bristles at such criticisms. She thinks that the presence of microbes in the brain must indicate a role for them, and she and others think they have good evidence that viruses are a linchpin in Alzheimer’s. “Most of us always acknowledged that amyloid was a very important feature of Alzheimer’s — but it is just not the cause,” she says.
Several microbes have been proposed as triggers of Alzheimer’s, including three human herpes viruses and three bacteria: Chlamydia pneumoniae, a cause of lung infections; Borrelia burgdorferi, the agent of Lyme disease; and, most recently, Porphyromonas gingivalis, which leads to gum disease. In theory, any infectious agent that can invade the brain could have this trigger role (there’s no good evidence, however, that SARS-CoV-2, the virus behind COVID-19, has this ability).
Most groups in this field have a favoured microbe, and two attention-grabbing papers in 2018 examined the role of the herpes viruses. One, from the group of Joel Dudley at the Icahn School of Medicine at Mount Sinai in New York City, analysed huge tranches of data on genes, proteins and tissue structure generated from nearly 1,000 post-mortem brains available in various databases. The team looked for telltale signatures of viruses in brain tissue — snippets of genes or proteins specific to herpes — and concluded that levels of human herpes virus 6A (HHV-6A) and human herpes virus 7 were higher in people who had Alzheimer’s disease than in controls2.
But other researchers, including virologist Steven Jacobson at the National Institute of Neurological Disorders and Stroke in Bethesda, Maryland, whose team studied a sample of more than 1,000 post-mortem brains, failed to replicate Dudley’s finding3.
And despite the impressive number of individual brains in Dudley’s study, the results are correlative. The source of the data is worrisome, too, says Michael Heneka at the German Centre for Neurodegenerative Diseases in Bonn. The brains of people with Alzheimer’s are in bad condition before death, and tissue breaks down further before autopsy; microbes could easily leak into them in the last days of life or after death. “We can’t make too many assumptions from post-mortem material about the pathogenesis of a disease which has a trajectory of approximately three decades,” he says.
Dudley’s paper came hot on the heels of a decade-long study in Taiwan, which followed more than 8,000 people who were diagnosed with herpes simplex virus, and compared them with a control group of 25,000 who had not received the same diagnosis. The group of people with herpes had a 2.5-fold increased risk of developing Alzheimer’s disease, but that increase was almost eliminated in those who received aggressive drug treatment4.
Even before this recent uptick in the theory’s prominence, the idea that infections might somehow provoke Alzheimer’s had enough traction for researchers to launch a clinical trial. In 2017, a team at Columbia University in New York City began to test whether the antiviral drug valacyclovir could slow cognitive decline and amyloid-plaque formation in people with mild Alzheimer’s disease who had also tested positive for antibodies to herpes simplex virus. Results are expected in 2022.
Burden of proof
When human studies provide only correlation, researchers often turn to animal experiments to look for cause. But animal models of Alzheimer’s aren’t perfect; mice, for instance, don’t develop the hallmark plaques as they age, unless they are genetically engineered to produce them. The widely used 5xFAD transgenic mouse expresses five relevant mutations in genes that code for the pre-amyloid protein and one of the enzymes that chops it into amyloid-β. These mice express the genes at super-high levels, and they start to develop plaques when they are only two months old.
Neurogeneticist Rudolph Tanzi and his colleagues at Massachusetts General Hospital in Charlestown used the 5xFAD mouse model to investigate a long-shot idea that emerged one Friday afternoon in 2008, when the department’s traditional ‘beer hour’ — also known to staff and students as ‘attitude adjustment hour’ — was under way.
Tanzi had been looking for Alzheimer’s risk genes in some new human genomics data and was puzzled to see a gene for CD33, a protein widely expressed in the innate immune system, pop out. He went next door to ask his friend and colleague Rob Moir what he thought about the odd idea that the innate immune system could throw up a candidate Alzheimer’s gene.
Moir, a neuroscientist, was busy surveying what was new in the general life-sciences literature, and had come across a paper about antimicrobial peptides, which are found in many innate-immunity pathways. “Dude, look at this,” he called to Tanzi. His computer showed a table describing the peptides, all of them of similar length to amyloid-β and with some similar properties. “Do you think amyloid-β could be an antimicrobial peptide?” he asked. Tanzi didn’t hesitate. “Let’s test it!”
Moir ran with the idea. “He was like a dog with a slipper, he wouldn’t let go,” recalls Tanzi.
At this point, no one had thought much about whether amyloid-β might have a specific role of its own, despite the fact that it is highly conserved across species — a strong indicator of biological usefulness. The sequence is at least 400 million years old, and is present in around two-thirds of all vertebrates. Maybe it was more than just a bad guy, they speculated. Maybe it had a good-guy function, trapping microbes that find their way into the brain and stopping them from causing disease. This system might go wrong when the brain ages and loses its ability to efficiently clear the amyloids.
Tanzi, who had majored in microbiology, asked postgraduate student Stephanie Soscia to quickly look at whether amyloid-β could kill eight common disease-causing microorganisms in a test tube, including Streptococcus pneumoniae and Escherichia coli. It could, she found — at least as effectively as known antimicrobial peptides could.
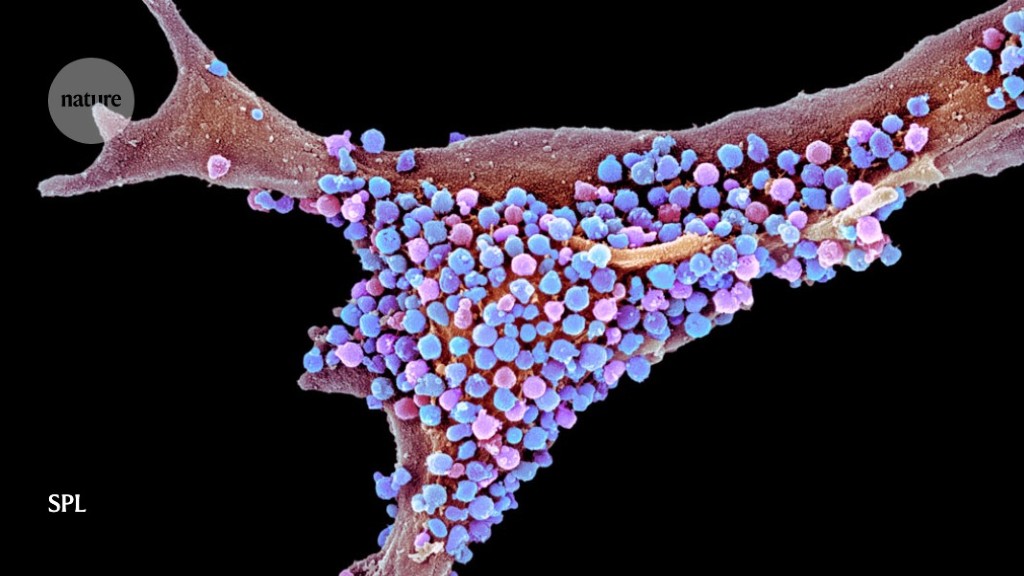
They raced that fact into press5 in 2010, and over the following years, Moir presided over a series of more-thorough experiments to probe what they were now calling their antimicrobial protection hypothesis. They injected the bacterium Salmonella typhimurium directly into the brains of plaque-making 5xFAD mice, and found that they survived longer than non-transgenic, plaque-less mice. They found similar results in nematode worms, using the pathogenic fungus Candida albicans. In both cases, amyloids formed sticky nets that engulfed and disarmed the pathogens6 (see ‘How microbes could seed plaques’).
Then the team turned its attention to herpes viruses, which had emerged as the human pathogens most frequently linked with Alzheimer’s disease. They injected HSV1 into the brains of young 5xFAD mice and of normal mice. Within three weeks, the brains of the transgenic mice were dotted with amyloid plaques. When the team repeated the experiment with a lethal dose of HSV1, the transgenic mice lived longer than the controls — and plaques appeared in their brains within a remarkable two days7. “It was an amazing thing to see,” says Tanzi.
HSV1 is so widespread that well over half of people worldwide harbour it in their bodies. But Moir also wanted to test the effects of HHV-6, which is found in up to 10% of healthy brains — albeit often at low levels, and with unknown effect. Mice are resistant to HHV-6 infection, so Moir’s team probed the effects of the virus in a 3D culture of human neural cells that models some aspects of Alzheimer’s disease. Normally, this mini brain organoid begins to accumulate amyloid plaques and tau tangles after six weeks in culture. But, as the researchers had seen with mice, plaques appeared just two days after they added the virus7.
Moir and Tanzi went on to investigate the impact of herpes viruses on tau-tangle formation in the organoids, and whether tangles could block the spread of viruses down neurons. Moir died in December 2019 after a short illness, but Tanzi says his group is still pursuing this line of work.
The upshot of his proof-of-concept experiments so far, he says, is that “if you are making amyloid-β, you survive infection better”. But he admits that actual proof — seeing an infection trigger the amyloid cascade to cause disease — is a long way off. “We haven’t seen the smoking gun.” And no one yet knows whether amyloid-β’s antimicrobial properties are actually deployed as part of a normal physiological process in people, he says, or how significant they would be in the general palette of defence mechanisms in the brain. Infection could be one way of striking the match that leads to the fire of Alzheimer’s, just as rare genetic mutations do.
Aware that whatever strikes the match at the onset of the disease might no longer be around by the time the person dies, Tanzi’s laboratory is developing techniques to isolate and analyse individual plaques to see if traces of microbe are trapped inside. It’s a sort of archaeological dig, he says.
Supporting studies
Tanzi’s work has not yet been reproduced independently, but other experiments have provided circumstantial support for the antimicrobial protection hypothesis. For example, scientists at the biotech firm Genentech in South San Francisco, California, showed that a mutation in a gene known as PILRA, which is expressed in various immune cells, is associated with a reduced risk of Alzheimer’s8. The gene makes a protein that helps herpes and other viruses to enter neurons, and the researchers say the mutation might prevent this entry.
And most intriguingly, a 2020 paper9 from the lab of chemical biologist Yue-Ming Li at Cornell University in New York City provides a mechanism that might link neuroinflammation to amyloid-β production. Li’s team found that a protein called IFITM3 gets turned on when viruses make their way into the brain. The protein binds to one of the amyloid-making enzymes, called γ-secretase, and increases amyloid production.
Li and his team looked at specimens from brain banks, and found that expression of the IFITM3 gene rose with age. It was also higher in brains from people with Alzheimer’s disease than in those of controls. What’s more, in experiments with cultured brain cells, they found that an inflammation-boosting molecule, a cytokine called interferon, increased levels of both IFITM3 and amyloid-β (in human brain samples, too, wherever they found more IFITM3, they found more interferon). This all suggests, they say, that the protein could act as a go-between for inflammation and the amyloid-making process.
Li is now investigating whether IFITM3 could become a biomarker that would help in deciding which patients might be recruited to clinical trials of anti-inflammatory therapies or drugs that target γ-secretase. He is also probing whether the protein could become a useful target for drug development.
The results are “a big step forward”, says de Strooper, because they reveal the kind of cascade that characterizes many complex diseases, including cancer. The process “can be triggered either by mutations that cause familial Alzheimer’s disease which lead to more amyloid which drives inflammation, or by an infection which leads to inflammation which drives overproduction of amyloid peptides”, he says.
If this holds true, he says, it would have important implications for the treatment of Alzheimer’s, because blocking the production of amyloid-β might mean that infections suddenly pose a bigger threat to the brain. “But this is entirely speculative, and it depends how important amyloid-β might turn out to be in the global defence line of the brain.”
Some researchers are still sceptical that infections have a major role in Alzheimer’s. Neuroscientist John Hardy at University College London, who shared the 2018 Brain Prize with de Strooper and others for work on Alzheimer’s, says he’d “bet £5 but not £500” on the antimicrobial protection theory being right. “But I don’t think it is going to be provable, and I don’t think there is much left that needs to be explained about Alzheimer’s beyond the genetics,” he says. And neuroscientist Tara Spires-Jones at the University of Edinburgh, UK, says that although the data so far allow for the possibility that infection seeds some cases of Alzheimer’s by creating inflammation, the normal process of ageing could also be an explanation. Ageing, she points out, is the biggest risk factor for developing Alzheimer’s. “In my personal opinion, the general inflammation in the brain that comes with ageing is more likely to be the cause.”
Given the right models, however, some scientists think that the infection theory could be provable, even if it might be difficult to show what proportion of Alzheimer’s cases were sparked by a microbe. Jacobson is enthralled by the new possibilities, and is hoping to develop a marmoset model to test the infection theory, because this small primate emulates the Alzheimer’s pathology in humans more exactly than other models do. Tanzi is planning to use a mouse whose amyloid genes have been swapped for their human equivalents, and which therefore expresses human amyloid-β at normal physiological levels. Another key step will be for independent labs to replicate existing findings.
As for Norins’s award, 40 applicants have so far submitted work in the hope of walking away with the cash prize in March, when the challenge results will be announced. Norins is aware of the enormity of the task. Proof that a germ causes Alzheimer’s, he says, will be “the hardest evidence to provide”.