Shortening tuberculosis (TB) treatment by optimizing antibiotic dosing may be possible using data acquired from PET/CT scans. In a first-in-human study, scientists successfully measured antibiotic concentration–time profiles in patients with pulmonary TB, an important first step towards the use of PET scans as a tool to optimize TB treatment (Nature Med. 10.1038/s41591-020-0770-2).
Treatment of TB typically requires the patient to take prescription drugs for six or more months. Currently, the typical daily dose of rifampin, a first-line TB drug, is 10 mg/kg; but doses of up to 35 mg/kg per day are safe in adults. Higher drug concentrations at infection sites could more effectively kill pathogens, making treatments shorter, and reducing the risk of failure, relapse and death.
Effective treatment of infections depends upon achieving adequate levels of the antibiotic at the infection sites, where the microbes reside. However, due to the difficulties of directly sampling infected tissues, information on rifampin levels at infection sites has only been available from TB patients with refractory disease, where resections are performed to remove the infected areas.
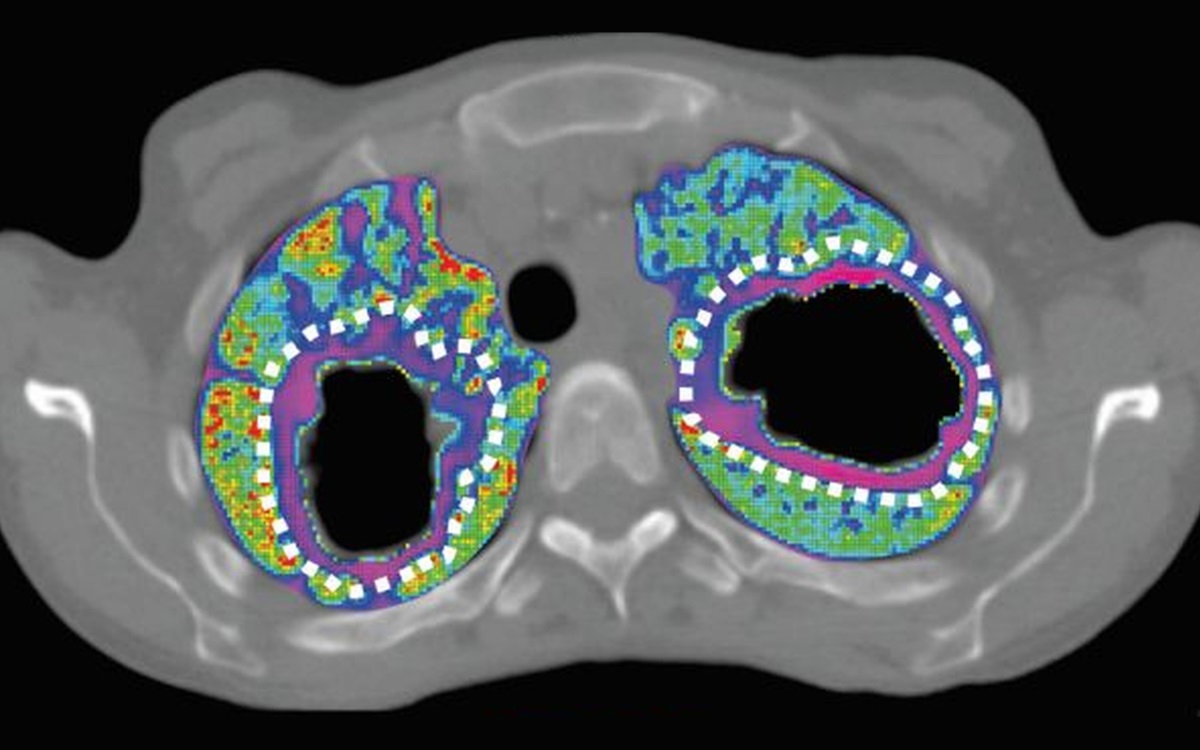
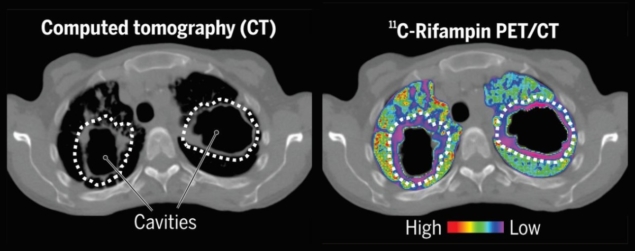
Principal investigator Sanjay Jain and lead author Alvaro Ordonez, of the Center for Tuberculosis Research at Johns Hopkins University School of Medicine, overcame this challenge using PET/CT with 11C-rifampin, a radiolabelled analogue of rifampin. They acquired area under the concentration–time curve (AUC) data from microdose dynamic 11C-rifampin PET/CT scans. The PET data were then used to determine concentration–time profiles of rifampin at infection sites, which provided a “translational bridge” allowing for pharmacodynamic modelling, according to the authors.
The team performed a multi-institutional study in 12 patients (nine men and three women, aged 19 to 77 years) newly diagnosed with rifampin-susceptible pulmonary TB. Half of the patients had pulmonary cavitation, thick-walled abnormal spaces in the lung, and all had been receiving treatment for at least 10 days prior to their PET/CT scan. The researchers made a total of 1221 measurements, including 473 from infected lung lesions and 748 from uninfected areas including the lung, brain, liver and plasma.
The researchers used the PET data to calculate the tissue-to-plasma AUC ratio for each pathologically distinct lesion using a plasma pharmacokinetics model. The model described the distribution of 11C-rifampin into affected and unaffected lung regions using the concentration–time profiles obtained from the PET data. CT results showed that pulmonary lesions were heterogeneous in different lung regions of a patient. 11C-rifampin exposures in pulmonary TB lesions were low, spatially compartmentalized and demonstrated between- and within-patient variability. The 11C-rifampin AUC tissue-to-plasma ratios were lowest in cavity walls, compared with other TB lesions or unaffected lung.
“This was eye-opening, since cavities are known to harbour the largest number of mycobacteria within infected lungs,” said Ordonez. “Cavitary TB patients are also more likely to fail treatment and spread the disease to other individuals. Rifampin is not getting where we need it most.”
To confirm the human findings, the team performed 11C-rifampin PET/CT on rabbits infected with cavitary TB that closely replicates human pathology, before and after 30–50 days of anti-TB treatment. The PET/CT images also demonstrated limited and spatially compartmentalized 11C-rifampin exposure in TB lesions, with the lowest levels in cavity walls. Post-mortem mass spectrometry in these infected rabbits showed a similar trend to the PET data.
The researchers used the PET data to develop an integrated pharmacokinetics model to predict the 11C-rifampin plasma and tissue concentrations in all patients, and the intra-lesional drug exposure in patients who received daily oral rifampin doses of 10–50 mg kg. With these data, they were able to predict accelerated cure rates based on different rifampin doses. Their results suggest that increasing the dose of rifampin to higher, yet safely tolerated levels, could reduce the treatment course in most TB patients from six to four months.
“This non-invasive approach to measure intralesional pharmacokinetics of antibiotics can also be used in other infections like methicillin-resistant Staphylococcus aureus, or MRSA, which is often treated with long courses of rifampin” Jain says. “We would finally be able to determine the most effective doses of specific drugs in specific patients.”